Brain disorders caused by mutations in single genes often present unexplained large symptom diversity. The new study shows that non-linear genetic interactions between two or more functionally related genes, STXBP1 and SNAP25, causes extreme phenotypic diversity. The study is published in Molecular Psychiatry.
Brain disorders caused by large effect mutations in single genes often present unexplained large symptom diversity, even among carriers of the same mutation. In the new study, a collaboration between FGA and the university of Copenhagen, the authors examined genetic interactions as a possible explanation for this diversity for SNAREopathies, a group of common neurodevelopmental disorders caused by de novo genetic variation in genes that together drive secretion of chemical signals in the brain. SNAREopathies are characterized by a striking phenotypic diversity, including different types/degrees or absence of seizures, developmental delay and intellectual disability.
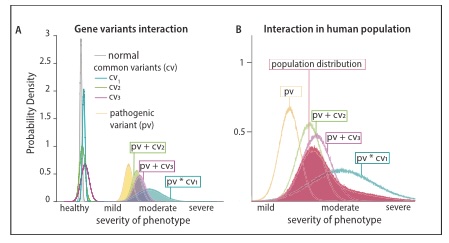
The authors tested the hypothesis that large phenotypic diversity is caused by non-linear genetic interactions between two or more functionally related genes by combining validated SNAREopathy mouse models and comparing phenotypic diversity between single and double mutants at the synaptic, network, system and behavioral level. Single Stxbp1 and Snap25 mutant animals showed EEG- and motor abnormalities, but no seizures, as reported before. In contrast, double mutants exhibited extreme diversity in seizure phenotypes. Some mice had lethal generalized seizures, frequent and complex epileptiform EEG activity and thalamic hyper-excitability as indicated by increased cFos staining, while other mice of the same genotype showed no detectable abnormalities, no increased cFos staining and a normal life span. The surviving double mutant mice showed phenotypes not more severe than single mutants at the synaptic, network, and behavioral level.
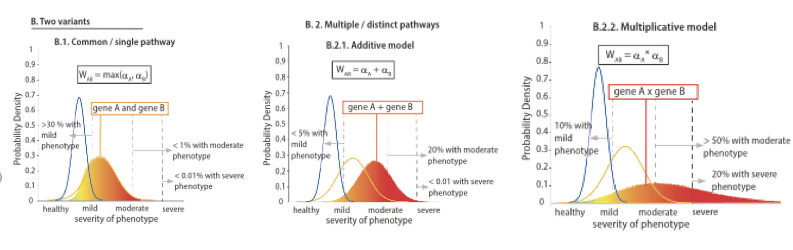
The authors also present a theoretical framework to quantitatively explain the observed non-linear effects and extrapolate the conclusions to symptoms diversity in human patients. This study provides a proof of concept for how modifying genes in the patient genome enhance phenotypic diversity.
The study can be found here.